You have no items in your shopping cart
Acetone
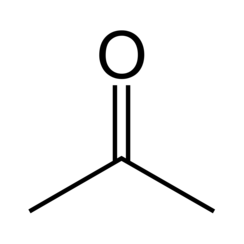
Acetone or propanone is an organic compound with the formula (CH3) 2CO. It is the simplest and smallest ketone. It is a colorless, highly volatile and flammable liquid with a characteristic pungent odor.
Acetone is miscible with water and by itself serves as an important organic solvent in industry, the home, and the laboratory. In 2010, approximately 6.7 million tons were produced worldwide, mainly for use as a solvent and for the production of methyl methacrylate and bisphenol A. It is a common building block in organic chemistry. Well-known household uses of acetone are as the active ingredient in nail polish remover and as a paint thinner. It has Volatile Organic Compound (VOC) exempt status in the United States.
Acetone is produced and disposed of in the human body through normal metabolic processes. It is normally present in blood and urine. People with diabetes produce it in greater quantities. Reproductive toxicity tests show it has a low potential to cause reproductive problems. Ketogenic diets that increase ketone bodies (acetone, β-hydroxybutyric acid and acetoacetic acid) in the blood are used to control seizures in infants and children with refractory epilepsy.