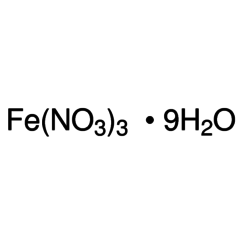You have no items in your shopping cart
Iron(III) nitrate
€12,08
Excl. tax
€28,14
Excl. tax
Iron(III) nitrate, or ferric nitrate, is the chemical compound with the formula Fe(NO3)3.
Applications
In the chemical laboratory
Ferric nitrate is the catalyst of choice for the synthesis of sodium amide from a solution of sodium in ammonia:
- 2 NH3 + 2 Na → 2 NaNH2 + H2
Certain clays impregnated with ferric nitrate have been shown to be useful oxidants in organic synthesis. For example, ferric nitrate on Montmorillonite—a reagent called "Clayfen"—has been employed for the oxidation of alcohols to aldehydes and thiols to disulfides.
Other applications
Ferric nitrate solutions are used by jewelers and metalsmiths to etch silver and silver alloys.