You have no items in your shopping cart
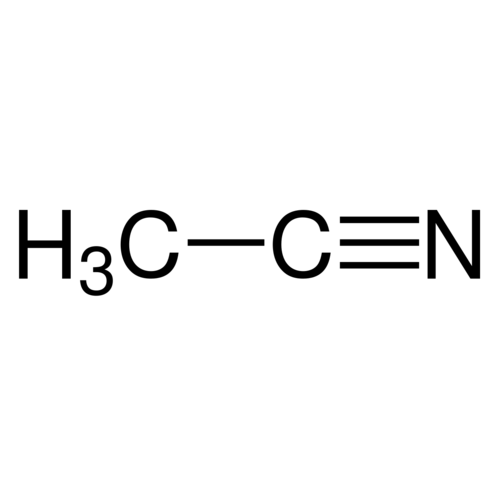
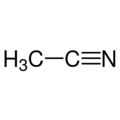
Acetonitrile 99.9+% Ultra Pure
- Buy 2 and save 5%
- Buy 6 and save 10%
What is Acetonitrile?
Acetonitrile is a colorless organic solvent with a boiling point of 82 degrees Celsius. It is the simplest natrile and is widely used both in the laboratory and in industry.
How is acetonitrile made and what is it used for?
Acetonitrile is obtained as a by-product of acrylonitrile production, therefore the trend of acetonitrile production generally follows that of acrylonitrile. The largest producers of this molecule in the United States are INEOS, DuPont, JT Baker Chemical and Sterling Chemicals. In 1992, 14,700 tons of acetonitrile were produced in the United States. In 2008, when the economic crisis led to a sudden drop in demand for acrylic fibers and resins, the production of acrylonitrile and acetonitrile fell and the price of acetonitrile increased fivefold in a few months. Acetonitrile is used as a solvent, particularly in the pharmaceutical industry, but also as a reaction intermediate in the production of many chemicals, from pesticides to perfumes.
It is a solvent of choice for testing an unknown chemical reaction. It is polar and the liquid domain is quite large which makes it handy. It easily dissolves a wide range of compounds due to the presence of non-acidic protons. Acetone exhibits the same properties but is more acidic and reactive towards bases and nucleophiles. In inorganic chemistry, acetonitrile is widely used as a ligand, abbreviated MeCN. For example, it is used in the form of PdCl2(MeCN)2 prepared by refluxing palladium dichloride in acetonitrile. It is a common solvent in cyclic voltammetry due to its relatively high dielectric constant. Acetonitrile is also commonly used in liquid chromatography where it serves as a mobile phase for the separation of molecules.
Buy Acetonitrile?
Acetonitrile Extra Pure from Laboratory Discounter is of high quality and for a low price. Available in packs from 100 ml to 25 litres. Delivered quickly and well packaged. So buy your acetonitrile at Laboratoriumdiscounter.nl!
Technical data
Cyanomethane, Methyl cyanide
Empirical formula C2H3N
Molar mass (M) 41.05 g/mol
Density (D) 0.79 g/cm³
Boiling point (bp) 81.65°C
Flash point (flp) 2 °C
Melting point (mp) -45.7°C
ADR 3 II
WCK 2
CAS no. 75-05-8
EC-No. 200-835-2
UN-No. 1648
Downloads
$$$$$
hazard statements
H225 Highly flammable liquid and vapour
H302+H312+H332 Harmful if swallowed, in contact with skin or if inhaled
H319 Causes serious eye irritation
Precautionary statements - prevention
P210 Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking
P280 Wear protective gloves/eye protection
Precautionary statements - response
P305+P351+P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuous ringing
Precautionary statements - storage
P403+P235 Store in a well-ventilated place. Stay cool